Single mutation linked to Crohn’s Disease
The paper: Murthy, A. et al. (2014) A Crohn’s disease variant in Atg16l1 enhances its degradation by caspase 3. Nature 506:546-562. doi:10.1038/nature13044
Subject areas: cell biology
Vocabulary:
Autophagy – “self eating” as defined by its Greek root words. In fact, autophagy is an orderly process by which nonfunctional or unneeded cellular proteins and other molecules are broken down into component parts small and versatile enough to be used in making new cell components.
—–
This article is a summary of a recent primary research paper intended for high school teachers to add to their general knowledge of current biology, or to supplement their lessons by showing students the kinds of projects that current biological research addresses.
—–
Crohn’s disease is a type of chronic inflammatory bowel disease that may affect as many as 700000 people in the United States. The most common symptoms, which range from mild to severe in different patients, are abdominal pain, fever, fatigue, loss of appetite, diarrhea, weight loss, and pain and/or blood when passing stools. Although its direct cause is unknown it is thought to be in part an autoimmune disorder, in which the immune system mistakes part of your body for a foreign infection and attacks it. Crohn’s has previously been shown to run in families, but this paper is the first to link a specific genetic change to increased likelihood of the disease.
The gene is ATG16L1, which encodes a structural protein that is needed to properly form autophagosomes. Autophagosomes are temporary organelles inside of a cell that act as cellular recycling centers: old or unneeded cellular components are dumped into them, and they are broken down into molecular building blocks that the cell can use for other things. It turns out that the mutation that changes “normal” ATG16L1 into a form that seems to be associated with Crohn’s disease is a change in just one nucleotide of the DNA, which then causes the ATG16L1 protein to have an alanine instead of a threonine at one particular point.
Now, if you have not had much molecular biology or biochemistry yet, you might wonder how one amino acid out of hundreds in one protein could contribute to a disease like Crohn’s. Most protein form specific complex shapes that are necessary for proper function. These shapes are held together in part by electrostatic interactions between positively and negatively charged areas of the proteins. The substitution of an alanine (A) for a threonine (T) at position 300 (T300A) removes a negative charge. What does that do? The authors showed that the mutation increases the sensitivity of the protein to cleavage by caspases. Caspases are a family of enzymes that chop up proteins, usually during apoptosis – an orderly breakdown and and eventual “suicide” of the cell. Caspase 3 was identified in this paper as the one responsible for ATG16L1 cleavage.
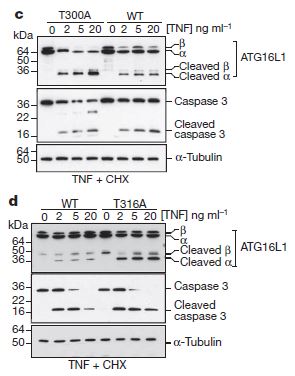 The figure at right demonstrates the increased cleavage. Looking at panel c, compare the leftmost four lanes at the level of “Cleaved alpha” with the rightmost four lanes. The left lanes are the mutant, and the right lanes are the normal or wild-type. The darker lines in the left show that activation of caspases by TNF (tumor necrosis factor – a cellular signal that is produced when the cell is under stress) increases the amount of cleaved ATG16L1 much more in the mutant than in the wild-type, though it does cleave both. In mice, which have a slightly different sequence for ATG16L1, the equivalent of T300A is T316A, or substitution at position 316 in the protein’s amino acid sequence. Here too in panel d, although the wild-type and mutant positions are switched from panel c, it is clear that there is enhanced cleavage of the mutant ATG16L1 protein.
The figure at right demonstrates the increased cleavage. Looking at panel c, compare the leftmost four lanes at the level of “Cleaved alpha” with the rightmost four lanes. The left lanes are the mutant, and the right lanes are the normal or wild-type. The darker lines in the left show that activation of caspases by TNF (tumor necrosis factor – a cellular signal that is produced when the cell is under stress) increases the amount of cleaved ATG16L1 much more in the mutant than in the wild-type, though it does cleave both. In mice, which have a slightly different sequence for ATG16L1, the equivalent of T300A is T316A, or substitution at position 316 in the protein’s amino acid sequence. Here too in panel d, although the wild-type and mutant positions are switched from panel c, it is clear that there is enhanced cleavage of the mutant ATG16L1 protein.
What does this have to do with any actual causes or symptoms of Crohn’s disease? When Murthy and colleagues generated mice carrying the T316A mutation, the mice did not have any developmental or inflammatory phenotypes, and intestinal cells also appeared normal at first. However, when the cells are stressed by glucose and amino acid starvation, there is a clear difference in ATG16L1 processing by caspases. Additionally, cultures of T316A-mutant mouse macrophages (a type of immune system cell) and T300A-mutant human macrophages are significantly less effective than wild-type macrophages at clearing a bacterial Yersinia enterocolitica infection. The T300A mutation is associated with Crohn’s disease affecting the ileum, and Yersinia often infects the ileum and moves through it to infect macrophages, so this observation could be an important link between gut bacteria and Crohn’s disease. In fact, after introducing Yersinia into the gut of intact mice, the T316A mutants were found to have significantly higher level of infection in the mesenteric lymph node than wild-type mice after 48 hours (figure at right, below).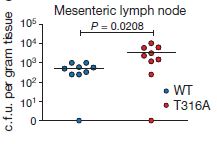
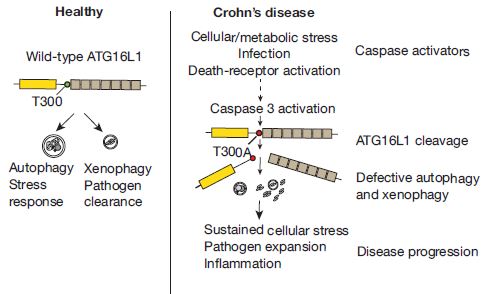
The researchers showed that a mutation that is linked to Crohn’s disease in humans could cause the kinds of cellular defects that could actually lead to some of the symptoms of Crohn’s. As the diagram shows, people with the normal ATG16L1 gene generate a normal autophagic or xenophagic response to stress or infection, respectively, forming autophagosomes to deal with the problem in a self-contained manner. However, if the ATG16L1 gene has the T300A mutation discussed in this paper, the autophagosomes are not properly formed because the protein is sensitive to cleavage by caspase 3, leading to continued stress or infection that spreads to neighboring cells, and gets progressively worse.
It is important to keep in mind though, that it is estimated that there are far more people carrying the T300A mutation (estimated at 50% of Caucasians) than there are people who have Crohn’s disease, so while this may certainly play a role in the development of Crohn’s, it is by no means the entire story.

No comments
Be the first one to leave a comment.